Psychedelic therapy is typically framed as a story of receptors, neurotransmitters, and neuroplasticity. Yet an emerging body of research suggests another protagonist may be quietly involved: the gut microbiome. Far from passive bystanders, microbial communities may influence how psychedelic compounds are metabolised, how inflammation is regulated, and why therapeutic responses vary so widely between individuals.
The modern psychedelic renaissance is frequently heralded as a revolution of the mind. As exogenous compounds like psilocybin, LSD, and MDMA migrate from the fringes of counter-culture into the rigour of clinical trials, the prevailing scientific narrative has remained stubbornly neurocentric. We speak of "rewiring" circuits and "resetting" the brain, focusing almost exclusively on the grey matter housed within the skull.
However, this brain-centric perspective overlooks a fundamental biological reality: the mind does not exist in a vacuum. The gut-brain axis, a sophisticated bidirectional communication network, is emerging as the silent architect of the psychedelic experience. To truly map the future of mental health, we must look beyond the cranium and into our microbial "mindscape." The efficacy of these substances may depend less on our thoughts and more on the complex ecology of our gastrointestinal tract.
The Bidirectional Loop and Systemic Inflammation
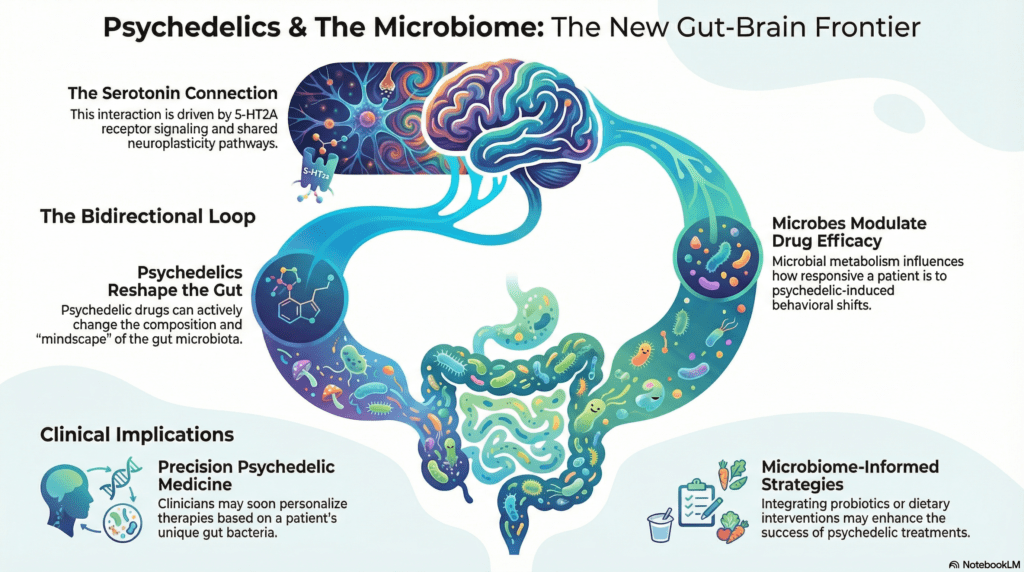
The interaction between psychedelics and the human body is not a linear event — it is a feedback loop. Research indicates that the pharmacological profile of substances like ketamine, ayahuasca, and psilocybin can actively reshape the composition of the gut microbiota. In turn, these microbial communities send signals back to the central nervous system, often using the "language" of systemic inflammation.
This loop challenges the traditional top-down model of psychiatric medicine. If the gut is in a state of dysbiosis, a microbial imbalance, it can trigger inflammatory pathways that blunt the brain's responsiveness to therapy. This suggests a decentralised model of health where the state of our internal bacteria dictates whether the body is receptive to healing or locked in a defensive, inflammatory posture.
Beyond the 5-HT2A Receptor
For decades, the standard explanation for the psychedelic experience has centred on partial agonism at the serotonin 2A (5-HT2A) receptor. This specific interaction is credited with driving the connectivity shifts and neuroplasticity that characterise therapeutic breakthroughs. While this receptor is undoubtedly a key gateway, it is only one chapter of a much larger physiological story.
Focusing solely on brain receptors ignores the broader context in which these compounds function. As Caspani et al. (2024) observe:
"These accounts fail to recognise that the gut microbiota, acting via the gut-brain axis, may also have a role in mediating the positive effects of psychedelics on behaviour."
Microbial Metabolism as a "Trip Filter"
Perhaps the most transformative finding is that microbial metabolism acts as a biological filter for psychedelic compounds. Before a substance like psilocybin can reach the brain to stimulate neuroplasticity, it is subject to the metabolic activity of our resident bacteria.
This microbial intervention determines the therapeutic window of a session. If the microbiome is not primed to process these exogenous compounds effectively, the intended rewiring of neural pathways may never occur, regardless of the dose. This metabolic gatekeeping explains the high degree of heterogeneity observed in clinical trials — why two individuals can receive the identical pharmacological intervention yet experience vastly different clinical outcomes. In essence, our microbes may decide how deeply we are permitted to heal.
The Rise of "Psychobiotics" and Precision Medicine
As we unravel these microbial mindscapes, the path forward leads to a new era of precision medicine in psychedelic therapy. The current one-size-fits-all dosing protocols often fail to account for the diversity of the human microbiome. The future of the field likely involves "psychobiotic" interventions — strategies designed to optimise the gut environment and ensure the brain is ready for change.
Future clinical practices may include:
Microbial Diagnostics: Pre-treatment screening of a patient's gut composition to predict drug efficacy before a session begins.
Psychobiotic Pre-conditioning: Utilising specific bacterial strains to reduce systemic inflammation and widen the therapeutic window.
Metabolic Priming: Tailoring diet and lifestyle to ensure microbial metabolism supports, rather than hinders, neuroplasticity.
Integrated Protocols: Combining psychedelic sessions with gut-first interventions to sustain the long-term antidepressant effects of the compound.
The Mindscape is an Ecosystem
We are beginning to understand that the human mind is not an isolated organ, but a biological ecosystem. Shifting our perspective from "mind over matter" to "microbial mindscapes" forces us to confront the fact that mental health is a full-body phenomenon.
If the gut-brain axis is a primary mediator of psychedelic action, it raises a profound question: are our daily choices regarding diet and lifestyle — the very factors that cultivate our microbiome — just as critical to our psychological resilience as the compounds currently under study?
To heal the mind, we may first need to tend to the garden within.