Introduction
The purpose of this study was to map the extent and limitations of primary human evidence concerning prenatal exposure to substances such as MDMA, psilocybin, and LSD. The researchers highlighted a persistent “pregnancy evidence gap” caused by the routine exclusion of pregnant individuals from clinical trials. This structural exclusion forces healthcare providers to rely on post-marketing observational data, case reports, and information from teratology services when counseling patients about potential risks.
Audio Summary
Key Findings
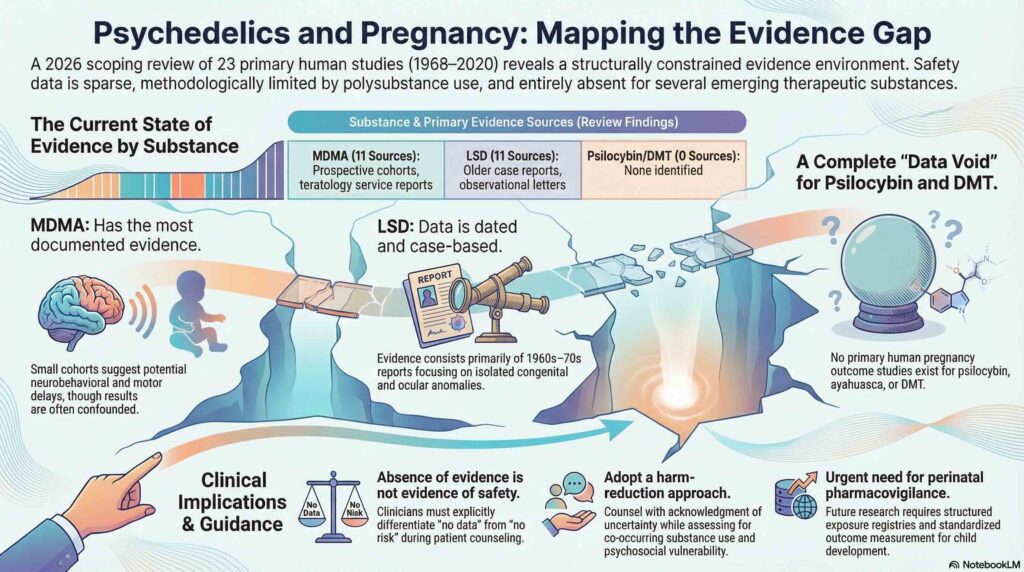
The scoping review identified only 23 primary human sources published between 1968 and 2020. The available evidence is heavily concentrated on MDMA (11 sources) and LSD (11 sources). Notably, the researchers found no eligible primary human pregnancy outcome studies for psilocybin, DMT, or ayahuasca. While a single case report exists for mescaline or peyote, the overall human evidence base for most psychedelics remains remarkably thin.
The evidence is characterized by significant heterogeneity and consists primarily of small cohorts and case reports. Several major threats to the validity of this data were identified, including a heavy reliance on self-reporting without biological confirmation through toxicology. Confounding by polysubstance use was also common, as many subjects in the studies used tobacco or cannabis concurrently. The LSD literature is notably older and provides historically important signals, yet it remains limited by these methodological constraints and inconsistent outcome definitions.
While the data do not establish causality, some signals were reported in the literature. These include reports of congenital anomalies in cases involving MDMA and LSD, as well as neurobehavioral and motor delays in infants within MDMA-exposed cohorts. However, the researchers emphasize that these observations must be interpreted within the context of the identified limitations, such as exposure misclassification and reporting bias.
Conclusion
The core takeaway for clinical practice is that the evidence regarding prenatal psychedelic exposure is sparse and structurally fragmented. A critical summary of the findings is that an “absence of evidence is not evidence of safety.” Clinicians should approach patient counseling by explicitly acknowledging these uncertainties, supporting harm reduction strategies, and focusing on individualized follow-up care for patients and their infants.