Introduction
The implementation of multi-model QSAR systems facilitates a proactive hazard assessment where a significant data gap currently exists. While compounds such as psilocybin and N,N-dimethyltryptamine (DMT) are advancing as clinical candidates for depression and anxiety, other analogs frequently circulate as research chemicals in recreational markets without the benefit of systematic human safety trials. This study bridges the gap by utilizing computational modeling to estimate systemic toxicity, organ-specific effects, and genotoxicity. These in silico results provide an objective framework for assessing hazards in the absence of comprehensive animal or human experimental series, offering a structured way to evaluate risk for compounds gaining medical and forensic prominence.
Audio Summary
Key Findings
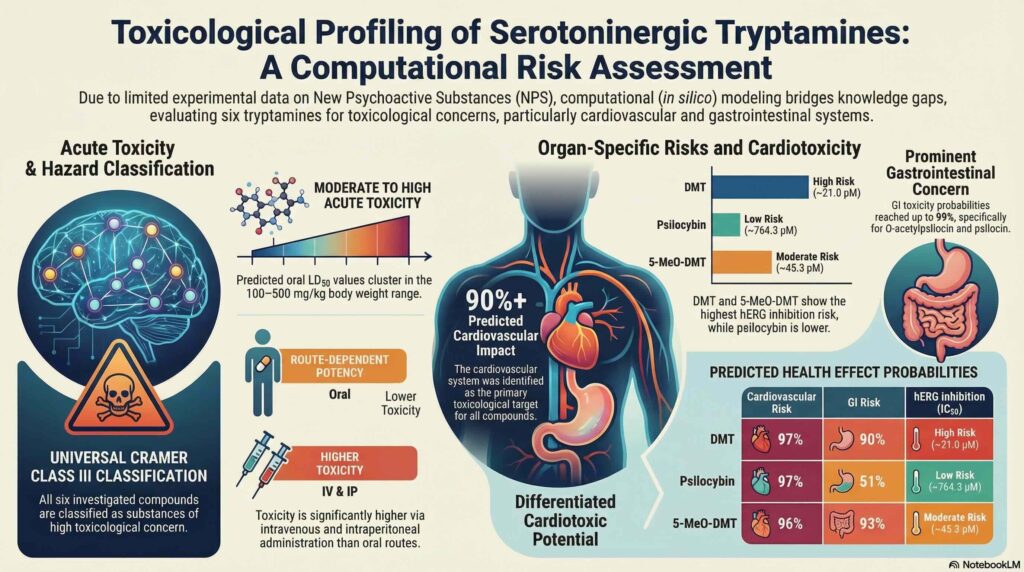
Analysis of the molecular structures identified specific toxicophores, namely the conjugated indole ring system and the protonatable side-chain amine, as primary structural alerts for toxicity. All six investigated compounds were assigned to Cramer Class III, indicating a high toxicological concern. While predicted oral LD50 values generally fell within the 100 to 500 mg/kg range, suggesting moderate to high acute toxicity, a senior assessment must account for route dependency. Parenteral administration exhibits a markedly higher hazard profile; for instance, the intravenous LD50 for psilocin in mice is predicted at 39 mg/kg, which is significantly more toxic than the corresponding oral estimates.
The cardiovascular and gastrointestinal systems were identified as primary target organs, with predicted effects at or above 90% across the series. Cardiotoxic potential was specifically evaluated via hERG inhibition, where DMT and 5-methoxy-N,N-dimethyltryptamine (5-MeO-DMT) exhibited the highest risk profile within the 20<IC50<45μM range. Specifically, DMT showed an IC50 of 21.0μM. In contrast, psilocybin demonstrated a significantly lower risk with an IC50≈760μM. These profiles are particularly relevant for O-acetylpsilocin (4-AcO-DMT, also known as psilacetin), which acts as a prodrug to psilocin and shares its shared toxicophoric core.
In terms of genotoxicity and endocrine activity, the results suggest a low risk for this chemical group. Most investigated tryptamines were predicted to be non-mutagenic via the Ames test. Furthermore, the compounds demonstrated a lack of activity regarding the estrogen receptor alpha (ER−α). All predicted LogRBA values remained below -3, which is the established threshold for non-binders. While certain models showed divergence for specific analogues, the consensus data indicate that these tryptamines generally carry a low risk for endocrine disruption or mutagenic activity.
Conclusion
These findings provide a structured evidence base for clinical management and forensic interpretation, especially in scenarios where human or animal data are absent. The results support treating these tryptamines as substances of significant acute toxicological concern based on computational predictions. This analysis serves as a critical tool for regulatory deliberations and the management of acute intoxications, ensuring that clinical and forensic practitioners can make informed decisions based on standardized hazard assessments.