Introduction
In the 21st century, human populations face widespread circadian disruption driven by the constant use of artificial light sources and various sleep disorders. This chronic misalignment between internal rhythms and the external environment has led to the emergence of the “circadian syndrome,” a concept that expands the definition of metabolic syndrome to include sleep disturbances and depression as core components. Notably, research suggests that the circadian syndrome is a stronger predictor for cardiovascular disease than the metabolic syndrome alone. This narrative review evaluates vitamin D as a bioactive compound that may play a significant role in synchronizing the circadian clock and mitigating the risks associated with metabolic and rhythmic imbalances.
Audio Summary
Key Findings
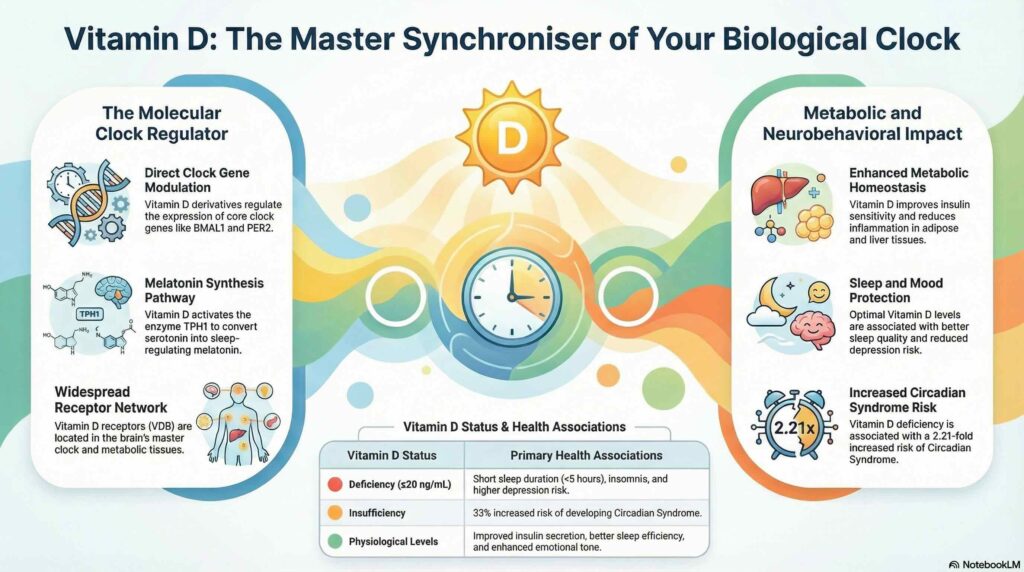
At the molecular level, vitamin D and its hydroxyderivatives influence core “clock genes,” such as BMAL1 and Per1. By interacting with transcriptional regulators like RORs and REV-ERBs, vitamin D helps to reset the phase and amplitude of the biological clock, facilitating the proper timing of cellular processes. A crucial molecular bridge to sleep health is the regulation of melatonin synthesis; the review details how vitamin D activates tryptophan hydroxylase 1 (TPH1), which converts serotonin to melatonin in the pineal gland during the evening hours.
The review further identifies a clear relationship between vitamin D deficiency and the development of metabolic syndrome features, specifically insulin resistance and impaired lipid metabolism. Furthermore, vitamin D deficiency is linked to the development of metabolic dysfunction-associated steatotic liver disease (MASLD), which is a core component of the circadian syndrome. Low levels of the vitamin are also strongly associated with poor sleep quality and the presence of depressive states, suggesting that vitamin D is vital for maintaining both metabolic homeostasis and emotional tone.
While observational data consistently link low vitamin D levels to circadian and metabolic disruption, results from interventional supplementation trials remain heterogeneous. Evidence from studies such as the VitDHiD research suggests that the response to vitamin D is individually sensitive, indicating that its impact on gene expression varies among participants. These variations highlight the complexity of how individuals respond to the vitamin and underscore the necessity for further personalized research.
Conclusion
Vitamin D serves as a promising regulator of both circadian rhythm and metabolic homeostasis, offering potential benefits for individuals living in environments characterized by significant light pollution. Within the scope of current evidence, it is clear that vitamin D influences the synchronization of the clock machinery with environmental cues, relying on its chronobiological action to facilitate the phase-shifting of physiological rhythms. However, future research must determine the optimal timing for vitamin D administration and account for individual susceptibility to ensure effective clinical application.