Introduction
This review was conducted to address the rising global incidence of acute cerebral infarction (ACI), which remains a leading cause of adult disability and mortality. While thrombolytic therapy with tissue-type plasminogen activator (tPA) is the established clinical intervention, its application is limited by a more than 4-fold increased risk of hemorrhagic transformation within the first 12 hours of treatment. These significant safety risks highlight the necessity for safer alternative or adjunctive neuroprotective strategies. The research explores whether Vitamin D can modulate the activity of HIFs, which are essential proteins that coordinate the cellular response to hypoxia, to support brain tissue survival and adaptation following an ischemic event.
Audio Summary
Key Findings
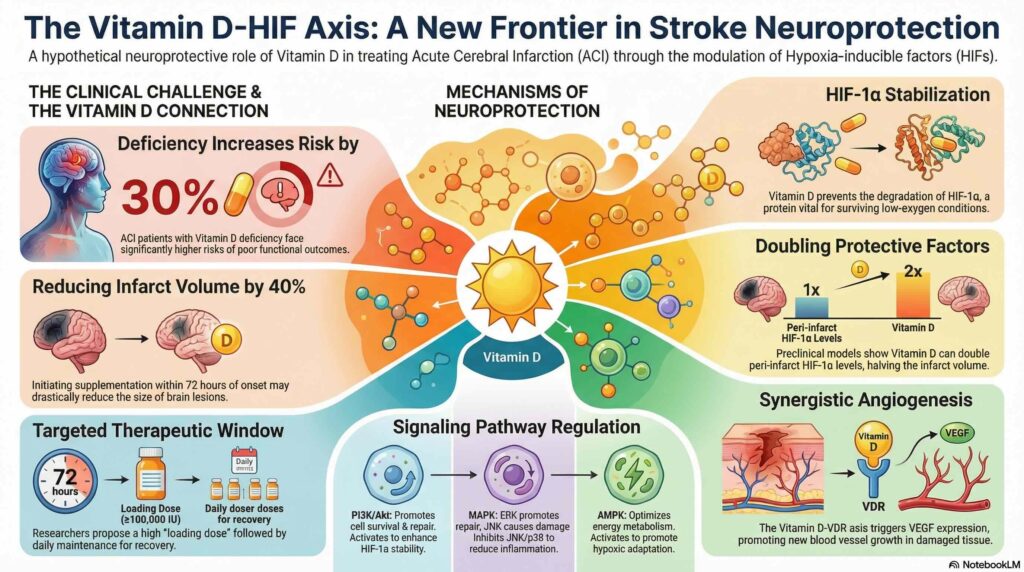
Preclinical and clinical evidence provides a strong rationale for investigating Vitamin D in the context of stroke recovery. In rat models of cerebral ischemia, Vitamin D supplementation has been shown to stabilize HIF-1α and reduce total infarct volume by as much as 50 percent. These results are supported by clinical cohort studies showing that Vitamin D-deficient ACI patients face a 30 percent higher risk of poor functional outcomes compared to those with sufficient Vitamin D levels.
The authors identify a biological “Vitamin D-HIFs axis” where Vitamin D provides neuroprotection by inhibiting the activity of prolyl hydroxylase (PHD). Under normal conditions, PHD facilitates the degradation of HIF-1α, but its inhibition by Vitamin D allows these protective factors to accumulate. This axis is further regulated through several key signaling pathways:
- PI3K/Akt and AMPK: Vitamin D activates these pathways to enhance the stability and functional activity of HIF-1α.
- NF-κB: Vitamin D inhibits this pathway to reduce the production of pro-inflammatory cytokines and limit tissue damage. By balancing these signals, Vitamin D helps maintain HIF expression within a range that promotes adaptive angiogenesis and cell survival without triggering excessive inflammation.
Based on these molecular insights, the authors propose a speculative therapeutic protocol for patients with Vitamin D deficiency. This hypothetical strategy involves the administration of a high loading dose (100,000 IU or more) followed by a maintenance dose of 6000 IU per day, initiated within 72 hours of symptom onset. The authors suggest that such an intervention could potentially reduce infarct volume by 40 percent or more by reinforcing the natural protective mechanisms of the HIF system during the critical post-stroke window.
Conclusion
The Vitamin D-HIFs axis represents a highly promising target for therapeutic development in the treatment of acute cerebral infarction. While the preliminary preclinical evidence is compelling, it is important to note that these strategies are currently hypothetical and exploratory. Further clinical validation through well-designed randomized controlled trials is essential to establish the safety and efficacy of these supplementation protocols in human populations.