Introduction
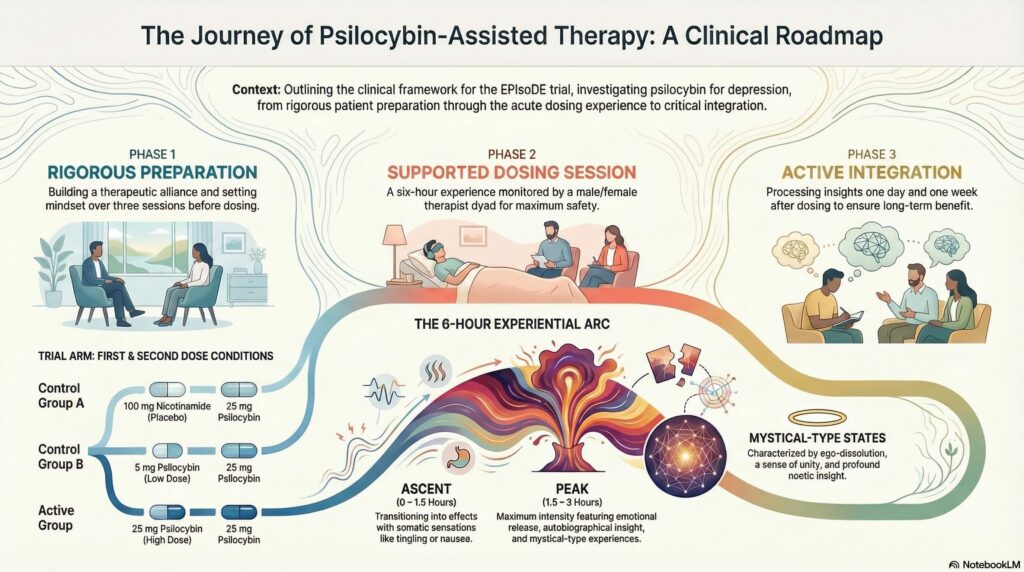
The EPIsoDE trial is a landmark Phase IIb study conducted in Germany, representing a pivotal return of psychedelic science to major German institutions like the Central Institute of Mental Health Mannheim and Charité – Universitätsmedizin Berlin. Investigating the efficacy and safety of psilocybin-assisted therapy for treatment-resistant depression (TRD), the trial utilizes the “Standard Protocol of Psychedelic Therapy.” This methodology places a high premium on the management of “Set and Setting,” where “Set” refers to the patient’s internal cognitive and emotional mindset and “Setting” refers to the curated physical and social environment. By exploring how this structured therapeutic framework influences patient safety and the subjective quality of the experience, the research seeks to define the parameters of a secure clinical intervention in a country where such research was restricted since the 1971 ban.
Audio Summary
Key Findings
The EPIsoDE trial employs a randomized, three-arm design featuring 25 mg psilocybin, 5 mg psilocybin, and 100 mg nicotinamide. The 5 mg dose serves as a sophisticated active control, providing transient physiological and psychedelic effects, such as alterations in mood, vision, and blood pressure, to maintain the integrity of the blinding for both participants and therapists. This low dose is capable of inducing detectable psychedelic phenomena, with prior data suggesting that a small percentage of participants may even reach the criteria for a mystical-type experience. Nicotinamide was selected as a further control to avoid the obvious skin flushing associated with niacin, thereby minimizing the risk of premature unblinding.
To ensure clinical safety, the protocol mandates a male-female therapist dyad to establish a secure experiential container for the patient. This gender-balanced pairing is designed to enhance the sense of psychological safety, particularly for individuals with histories of trauma. Lead therapists, who must be licensed physicians or psychotherapists with specialized training in TRD, provide a meditative and grounded presence throughout the session. This supportive framework is essential for managing the potentially intense noetic quality and paradoxicality of the psilocybin experience while maintaining strict professional boundaries and a non-directive therapeutic stance.
The experiential framework is further supported by eyeshades and a specialized Music Block System. To tailor the session to the patient’s shifting needs across the ascent, peak, descent, and return phases, therapists select one of five available 90-minute blocks for each specific phase of the six-hour dosing day. This selection process allows for an intuitive adjustment of the acoustic environment to match the patient’s emotional intensity and internal process. By minimizing external distractions and providing a curated auditory arc, the protocol facilitates the safe navigation of the psychedelic state, from challenging confrontations to profound insights.
Conclusion
As one of the first trials of its kind in Germany in decades, the EPIsoDE trial, conducted in collaboration with the MIND Foundation and Charité – Universitätsmedizin Berlin, signifies a major advancement in European psychiatric research. This therapist manual serves as the primary clinical guideline for the study, reinforcing the necessity of a rigorous supportive framework to manage the acute and often intense effects of psilocybin. The findings emphasize that the therapeutic context is an indispensable component of the treatment, suggesting that a carefully curated environment and a stable therapist dyad are as vital as the pharmacological agent itself. Ultimately, the study grounds psychedelic-assisted therapy within a modern, evidence-based medical paradigm.