Introduction
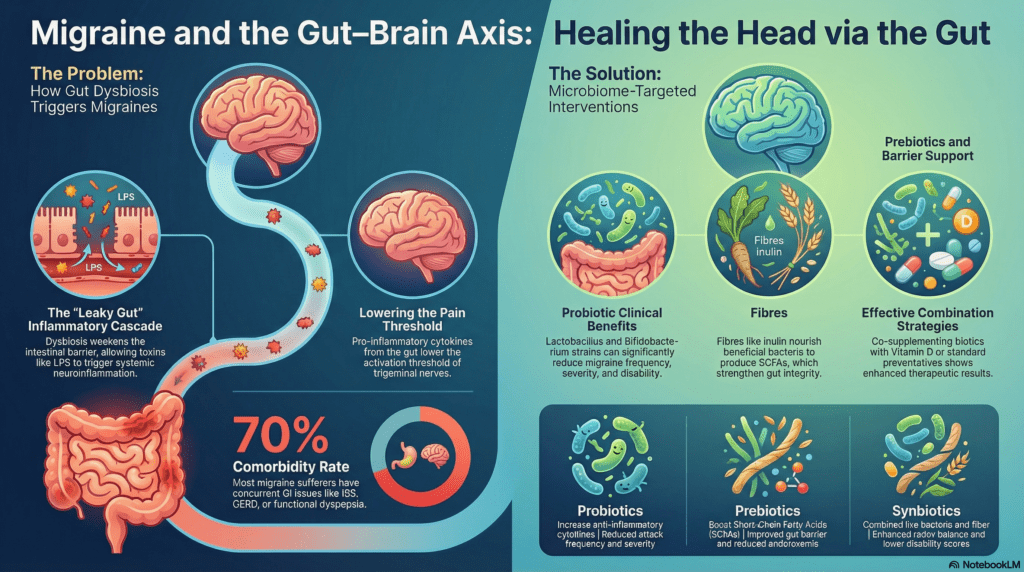
The bidirectional communication between the central nervous system and the gastrointestinal tract, known as the gut-brain axis, is a vital area of inquiry for the 70% of migraine patients who experience comorbid gastrointestinal disorders. Standard pharmacological treatments often prove insufficient for these patients, largely because they do not address the underlying “leaky gut” (increased intestinal permeability). When the intestinal barrier is compromised, the translocation of lipopolysaccharides (LPS) from Gram-negative bacteria into the systemic circulation activates Toll-like receptor 4 (TLR4). This process triggers a cascade of pro-inflammatory cytokines and chronic low-grade inflammation, which effectively lowers the activation threshold for neural sensitization. These biological mechanisms suggest that the gut serves as a critical upstream modulator for the neurovascular signals observed in clinical migraine trials.
Audio Summary
Key Findings
This research frames “biotics” not as simple dietary supplements, but as a paradigm shift in adjunctive therapy for precision-oriented care. Multi-strain formulations, particularly those containing Lactobacillus and Bifidobacterium, have demonstrated the ability to reduce attack frequency and severity by stabilizing the intestinal barrier. These strains promote the expression of essential tight junction proteins, including claudin, occludin, and zonula occludens-1 (ZO-1), which are necessary to prevent the systemic inflammatory signaling that leads to calcitonin gene-related peptide (CGRP) release and neurogenic inflammation.
When evaluating clinical responses, a significant differentiator is observed between adult and pediatric populations. Pediatric data show strong, consistent signals, with biotics significantly reducing attack frequency and disability scores when used as adjuncts to standard therapies like propranolol or sodium valproate. In contrast, adult data remain more heterogeneous. While primary headache endpoints in adults are not always reached, the documented improvements in quality-of-life and the management of gastrointestinal comorbidities represent a strategic therapeutic victory, offering clinical value even when traditional pain reduction is modest.
The review also identifies short-chain fatty acids (SCFAs), such as butyrate, as key candidate modulators that influence neuroinflammatory signaling. However, from a clinical perspective, it is important to note that while preclinical models for postbiotics are encouraging, human data for these metabolites remain limited. These findings suggest that biotics are best viewed as a means to decrease inflammatory tone and augment existing prevention strategies.
Conclusion
The evolution of migraine management necessitates a move toward precision-oriented care through standardized clinical trials and strain-specific interventions. While biotics provide promising signals for reducing the global burden of migraine, their effectiveness is highly dependent on the specific population and microbial formulation used. Ultimately, the gut-brain axis offers a biologically plausible and low-risk pathway for augmenting traditional migraine prevention. Future research must prioritize identifying specific responder profiles to fully integrate these interventions into clinical practice.