Introduction
The primary goal of this research was to evaluate whether anti-CGRP monoclonal antibodies (mAbs), which have demonstrated high success in migraine prophylaxis, offer similar benefits for cluster headache (CH). This investigation was driven by significant optimism regarding the role of CGRP in CH pathophysiology, yet regulatory bodies have reached different conclusions. For instance, galcanezumab is currently approved for episodic cluster headache (eCH) in the USA, but it lacks similar approval in Europe. To clarify these discrepancies, the researchers conducted a systematic review of clinical trials and real-world data to distinguish between efficacy under ideal trial conditions and effectiveness in everyday clinical settings.
Audio Summary
Key Findings
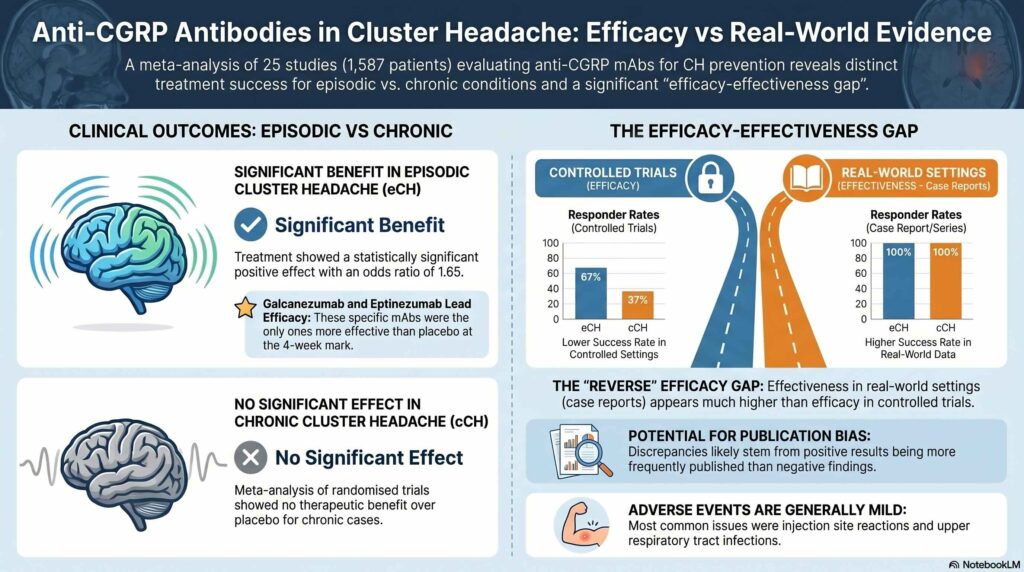
The meta-analysis of randomized, placebo-controlled trials demonstrated a statistically significant benefit for patients with episodic cluster headache (eCH), yielding an overall odds ratio of 1.65 (p=0.02). Specifically, 300 mg of galcanezumab and 400 mg of eptinezumab were more effective than a placebo in helping patients achieve a 50% or greater reduction in the frequency of their attacks. However, it is important to note that while responder rates were significant, some trials struggled to meet broader primary endpoints. For example, while galcanezumab 300 mg achieved a significant mean reduction in weekly attack frequency (p=0.04), eptinezumab failed to meet its primary endpoint regarding mean attack reduction, despite its success in responder rates.
In contrast, the researchers found that anti-CGRP treatments did not provide a statistically significant benefit for patients suffering from chronic cluster headache (cCH). The meta-analysis for this group resulted in an odds ratio of 1.07 (p=0.68), indicating that no CGRP antagonist was superior to a placebo in treating the chronic form of the disease.
The study also identified a “reverse efficacy-effectiveness gap” when comparing clinical trials to real-world data. While controlled trials showed moderate success, retrospective case reports often claimed success rates as high as 100%. The authors advise caution regarding these figures, attributing the discrepancy to potential publication bias where positive outcomes are more likely to be shared. Additionally, the researchers noted that selecting endpoints in cluster headache trials is difficult because attack frequency can change naturally over time, a factor that may obscure the true impact of a medication.
Conclusion
The current evidence indicates that while anti-CGRP mAbs show a beneficial effect for episodic cluster headache, they are not supported for use in the chronic form of the condition. These results emphasize the necessity for refined trial methodologies that can better account for the spontaneous changes and natural cycles inherent to the disease. While these findings clarify the current limits of CGRP-targeted therapy, interpretations must remain proportional to the data, as further research is required to optimize treatment approaches for different patient groups.