Introduction
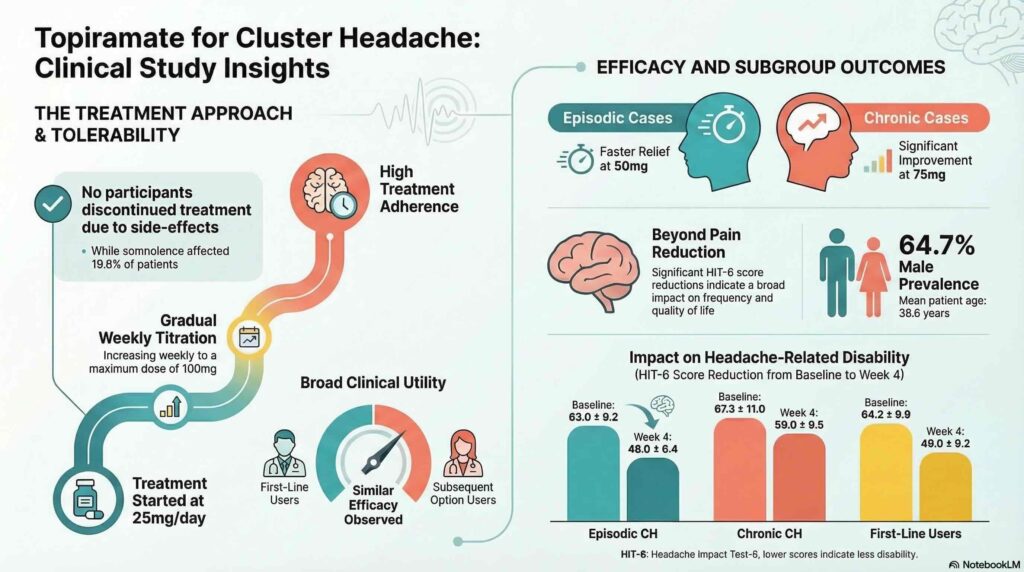
Establishing effective prophylactic regimens for cluster headache (CH) is a strategic priority in neurology because of the profound impact these attacks have on patient functionality and mental well-being. This prospective observational study followed a cohort of 51 adults, who were predominantly male (64.7%) with a mean age of 38.6 years. The research was designed to evaluate the effectiveness of topiramate monotherapy for both episodic and chronic CH, focusing on pain severity via the Visual Analog Scale (VAS) and headache-related disability through the Headache Impact Test (HIT-6). Topiramate has several pharmacologic mechanisms that may be relevant to headache prevention, including the blockade of voltage-dependent Na+ channels, the potentiation of gamma-aminobutyric acid (GABA), and the antagonism of the kainate subtype of the glutamate receptor. By documenting standardized scores throughout a titration period, the study sought to fill a research gap regarding quality-of-life areas not previously addressed in topiramate literature. These clinical observations provide a detailed view of patient response patterns in a real-world setting.
Audio Summary
Key Findings
The primary data outcomes demonstrate that topiramate monotherapy was associated with significant reductions over time in pain severity and headache-related disability scores. The marked decreases in HIT-6 and VAS scores indicate that the medication was associated with improvement in both pain severity and headache-related disability measures. By the end of the fourth week, utilizing a daily dose of 100 mg reached through gradual titration, the study recorded similar efficacy levels among patients regardless of whether they used topiramate as a first-line therapy or a subsequent option after prior treatment failure. However, a notable subgroup variance was observed: while both groups improved, the reduction in scores was significantly more pronounced in patients with episodic cluster headaches compared with those with chronic cluster headaches at the 100 mg target dose.
The safety profile was a critical highlight of the findings, with a 100% treatment retention rate despite a 19.6% incidence of somnolence. This high adherence rate suggests that the titration schedule effectively managed potential side effects. From a clinical management perspective, the study identified distinct response patterns that may be useful for patient counseling and titration management. Patients with episodic cluster headache typically showed notable improvement starting at a daily dose of 50 mg. In contrast, those with chronic cluster headache appeared to require around a 75 mg threshold before achieving a similar trajectory of relief. These dose levels should be interpreted as observed response patterns within this cohort rather than definitive dosing rules.
Conclusion
This prospective observational study suggests that topiramate monotherapy may be associated with meaningful reductions in pain intensity and functional disability for patients with both episodic and chronic cluster headaches. Topiramate may be considered a potential preventive option in selected patients, particularly where polypharmacy is a concern or where standard treatments such as verapamil are contraindicated or poorly tolerated. While these findings represent longitudinal associations that warrant further validation through larger randomized controlled trials, they support further study of a 100 mg daily target dose within controlled clinical settings.