Introduction
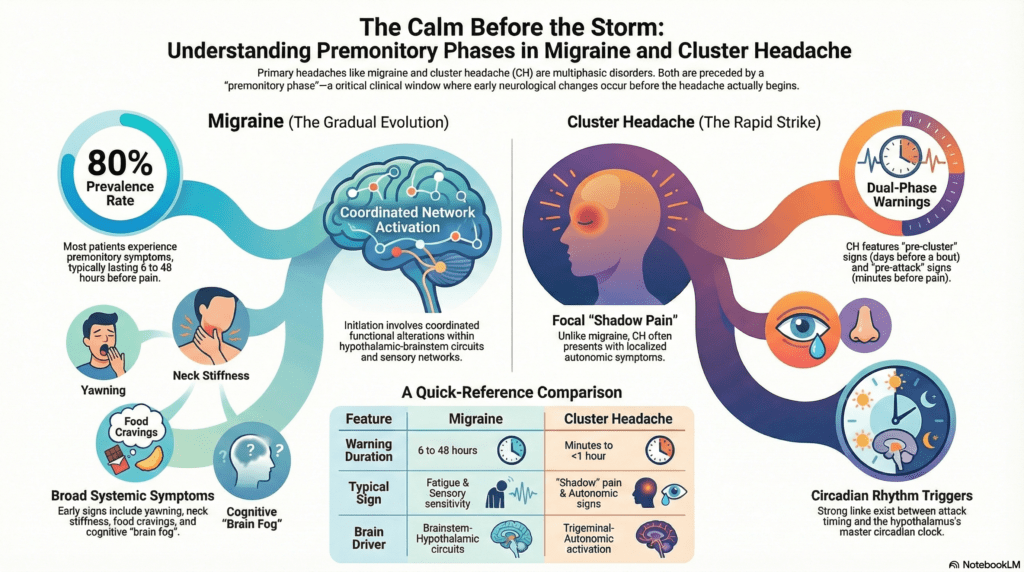
Characterizing the pre-pain phase is of strategic importance because it reveals a critical window where central nervous system alterations occur before a patient experiences overt pain. Defining the premonitory phase as the symptomatic period prior to headache onset allows clinicians and researchers to identify the earliest stages of the attack cycle. This study bridges the research gap between migraine and cluster headache (CH), examining how hypothalamic-brainstem circuits and trigeminal-autonomic networks coordinate to trigger these events. Recognizing these signals facilitates a transition from reactive care toward the specific neurobiological and clinical discoveries that underpin attack prevention.
Audio Summary
Key Findings
The investigation highlights distinct temporal and symptomatic profiles that differentiate the two conditions. In migraine, premonitory symptoms such as fatigue and sensory sensitivities typically evolve gradually over hours or even days. In contrast, cluster headache presents a more complex temporal structure, requiring a clinical distinction between pre-cluster symptoms, which emerge one to seven days before a new bout, and pre-attack symptoms, which arise rapidly within ten to sixty minutes of an individual attack. A hallmark of the CH pre-attack phase is shadow pain, defined as a dull, aching sensation that mirrors the laterality of the subsequent attack. While migraine involves broad functional alterations across the brain, CH is specifically associated with focal, ipsilateral posterior hypothalamic activation and pronounced circadian rhythmicity.
The neurobiological landscape further reveals significant mechanistic divergence regarding neuropeptide involvement. Although both disorders share hypothalamic-brainstem involvement, provocation studies show that while Pituitary Adenylate Cyclase-Activating Polypeptide (PACAP) triggers premonitory symptoms in migraine, it fails to induce pre-attack symptoms in CH. These findings shift the competitive landscape of headache treatment from reactive pain relief to pre-emptive intervention. Data from the PRODROME trial exemplify this shift, demonstrating that 46 percent of ubrogepant-treated events did not progress to moderate or severe headache within 24 hours compared to only 29 percent for placebo when treated during the prodrome. These developments underscore the potential of recognizing early-stage physiological markers as the basis for a paradigm shift in clinical neurology.
Conclusion
The identification of partially overlapping neurobiological substrates in migraine and cluster headache reshapes the current clinical paradigm by emphasizing the shared involvement of hypothalamic and brainstem circuits in attack initiation. Understanding these early changes in trigeminovascular sensory processing and trigeminal-autonomic network activation allows for the development of true pre-emptive therapy. By interrupting the progression of the attack before the onset of disabling pain, clinicians can move toward a more effective, personalized model of headache management.